Fully Reimbursed Medicinal Products in the Czech Health Insurance System
Fully reimbursed medicinal products play a key role in ensuring access to healthcare for patients in the Czech Republic. Their regulation is derived from Act No. 48/1997 Coll., on Public Health Insurance, which sets the conditions for their inclusion in the List of Prices and Reimbursements (SCAU). The goal is to ensure patients have access to treatment while optimizing public health insurance expenditures.
Full Reimbursement in Inpatient and Outpatient Care
Act No. 48/1997 Coll. stipulates that, during the provision of inpatient care, the following are fully reimbursed:
· medicinal products and foods for special medical purposes,
· individually prepared medicinal products,
· radiopharmaceuticals,
· transfusion products,
· medical devices,
· medicinal products for advanced therapy,
· tissues and cells.
These items are reimbursed in the least economically demanding version, and the insured person does not contribute to their payment.
In outpatient care, Annex No. 2 of the Act plays a crucial role in ensuring that at least one medicinal product in each therapeutic group is fully reimbursed by public health insurance. This guarantees patient access to treatment without co-payments, especially for chronic and severe diseases.
Full Reimbursement and Real Availability of Medicinal Products
However, full reimbursement of a medicinal product does not automatically mean its actual availability on the market. Data shows that only a portion of fully reimbursed medicines are genuinely available to patients, influenced by factors such as manufacturers’ supply decisions, demand fluctuations, and regulatory measures.
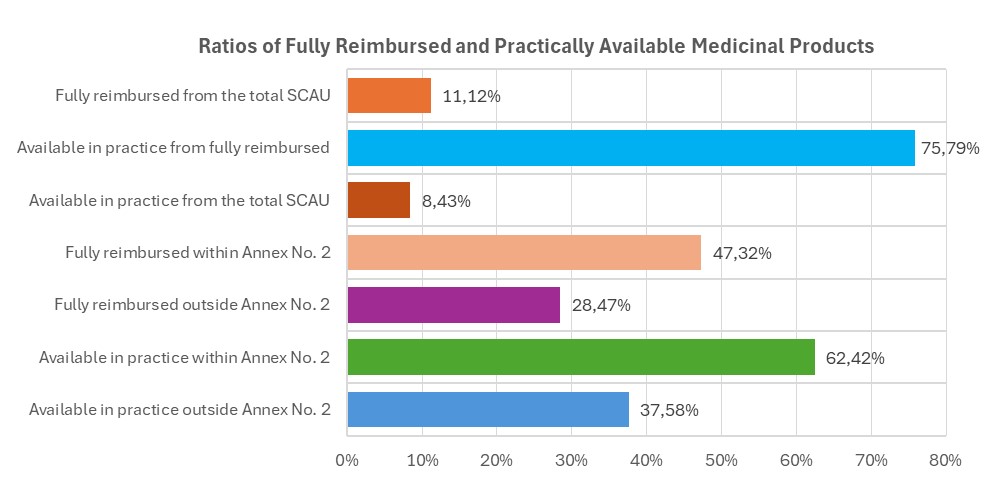
The list of medicinal products and foods for special medical purposes reimbursed by health insurance (SCAU250401) includes a total of 8,873 items, of which 987 are fully reimbursed. Out of this number, 748 products are available. The full reimbursement guarantee under Annex No. 2 of the Act applies to 467 of them, while 281 products are not included in Annex No. 2.
- Fully reimbursed products make up 11.12% of all items in the reimbursed medicines list.
- Available fully reimbursed products represent 75.79% of the total number of fully reimbursed products.
- Of the fully reimbursed items, 47.32% fall under Annex No. 2 of the Act.
- Outside Annex No. 2, 28.47% of fully reimbursed products exist.
- Available fully reimbursed products constitute only 8.43% of the total number of items in the list.
- Of the available fully reimbursed ones, 62.42% are from Annex No. 2.
- Outside Annex No. 2, 37.58% of the available fully reimbursed products remain.
Impacts on Patients and the Healthcare System
- Ensuring treatment availability – Annex No. 2 protects patients from the risk of a lack of fully reimbursed options in key therapeutic groups.
- Difference between reimbursement and availability – The fact that a product is fully reimbursed does not automatically mean it is present on the market.
- Prescription and economic impacts – Patients and doctors often prefer medications without co-payments, which influences prescribing patterns and healthcare system costs.
Conclusion and Future Outlook
Annex No. 2 of Act No. 48/1997 Coll. is a crucial tool for ensuring accessible healthcare. While it guarantees that a no-co-payment option will always be available in key drug categories, the actual availability of fully reimbursed products is influenced by additional factors that need to be monitored and addressed. In the future, it will be important not only to maintain the full reimbursement system but also to ensure that fully reimbursed medicinal products are genuinely available on the market for the patients who need them.

