Sekce:
Focused on
The Czech System for Pricing and Reimbursement of Innovative Medicines: What the Latest Data Show
Each European country approaches the pricing and reimbursement of medicines in its own way. The common objective, however, is to ensure that patients have access to medicines that meet EU-wide...
Pharmeca a.s.
03/11/2026
Sekce:
Daily overview
Selection from Decision-Making Practice - 4/2026
Tender Prices in Price Referencing.
Pharmeca a.s.
03/04/2026
Sekce:
Daily overview
Selection from Decision-Making Practice - 3/2026
Determination of Reimbursement to Ensure Availability.
Pharmeca a.s.
02/18/2026
Sekce:
Daily overview
Selection from Decision-Making Practice - 2/2026
Criteria Assessed for the First Similar Medicinal Product.
Pharmeca a.s.
02/04/2026
Sekce:
Focused on
Exchange Rates for Price Referencing after the Amendment
The amendment to the Public Health Insurance Act and its implementing decrees has introduced changes, effective from the beginning of the year, also in the way exchange rates are applied for price...
Pharmeca a.s.
01/28/2026
Sekce:
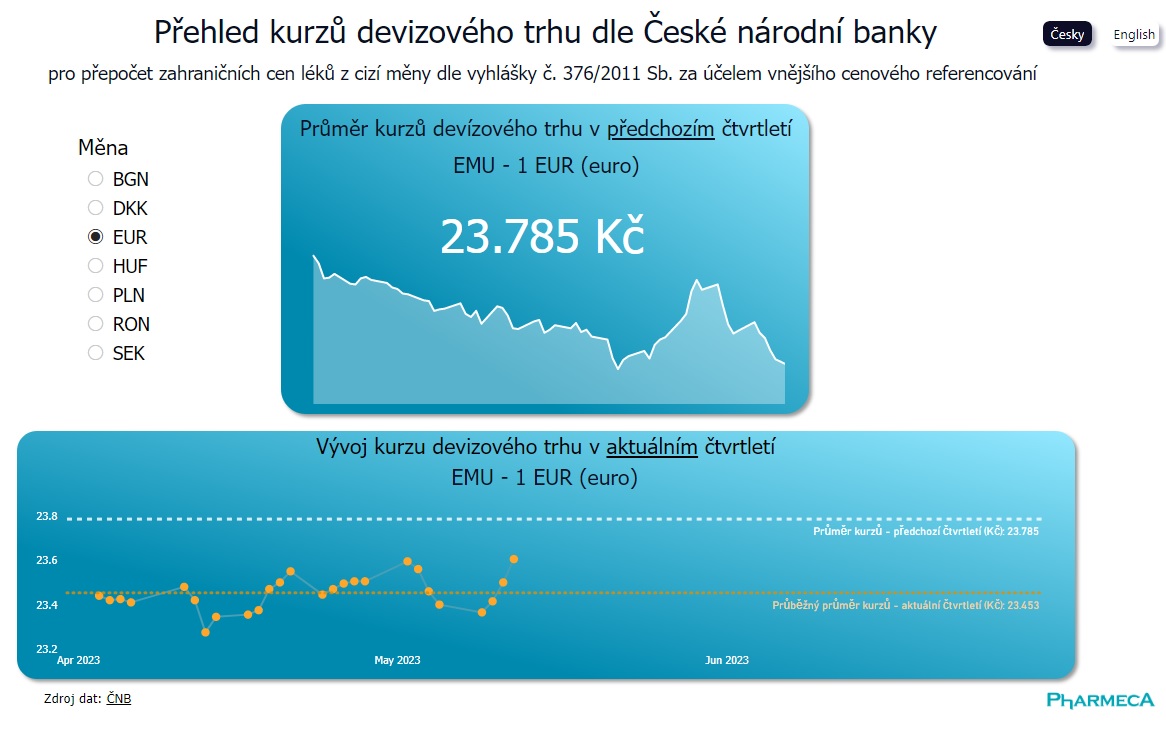
Data visualization
Exchange Rates for Price Referencing at a Click
The key information for conducting price referencing in administrative procedures for drug pricing and reimbursement is the average exchange rate from the previous quarter. Now, you can easily...

Pharmeca a.s.
01/28/2026
Sekce:
Daily overview
Selection from Decision-Making Practice - 1/2026
Ensuring a Fully Reimbursed Medicinal Product – Diseases of the Same Type
Pharmeca a.s.
01/14/2026
Sekce:
Focused on
Czech legislation
An up-to-date overview of selected Czech legal regulations regarding medicines and medical devices.
Pharmeca
12/30/2025

